Introduction
Lathyrism is a clinical condition associated with prolonged consumption of Lathyrus sativus (grass pea) and related species (L. cicera and L. clymenum).1 Its principal toxins include β-N-Oxalyl-α,β-diaminopropionic acid (β-ODAP), which causes neurolathyrism, and β-aminopropionitrile (BAPN), which is implicated in osteolathyrism and angiolathyrism.
Neurolathyrism is an irreversible upper motor neuron disorder characterized by degenerative changes in the spinal cord.2 Although its incidence has declined with improvements in food security and public health, it is endemic in areas such as Ethiopia, India, and Bangladesh, where reported prevalence is 7%–2.4%.3,4 Disease onset is typically acute or subacute, though insidious presentations have also been reported.5 However, sensory function is generally preserved. Clinically, the condition is categorized into 4 stages based on the severity of spastic paraparesis and mobility limitation. While progression may occur with continued toxin exposure, the disease course typically stabilizes once exposure ceases and is considered largely nonprogressive thereafter, with bladder and bowel function characteristically spared.5
β-ODAP acts as a glutamate analog at α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptors on long-projection corticospinal neurons, producing a distal axonopathy that predominantly affects the thoracolumbar cord.6,7 Poor nutritional status, particularly zinc deficiency, and heavy physical activity further increase the susceptibility to acquiring neurolathyrism.1 Although diagnosis is clinical and often one of exclusion, it should be considered in individuals with a predominant reliance on L. sativus as a major dietary staple over a prolonged period.
BAPN inhibits lysyl oxidase, leading to defective collagen and elastin cross-linking.1,2,8 This results in reduced bone toughness and trabecular thinning, as demonstrated in animal models showing significant decreases in fracture resistance.9 Some patients with chronic neurolathyrism also exhibit skeletal features of osteolathyrism, such as delayed apophyseal fusion and bowing of long bones, suggesting potential overlap in clinical features.8 Despite these observations, the mechanism underlying pathological fractures in lathyrism remains poorly defined.
In this case report, we discuss a young adult with neurolathyrism who sustained a low-energy, neglected femoral neck fracture and explore potential pathways contributing to increased fracture susceptibility by reviewing the existing literature.
Case Presentation
A 35-year-old male presented to our clinic with an inability to bear weight and significant left hip pain following a fall from a toilet seat 6 weeks before his visit. Initially, he sought treatment from a traditional bonesetter, but without success. His medical history includes neurolathyrism, diagnosed at the age of 10, which resulted in a bilateral limp and reliance on a walking aid.
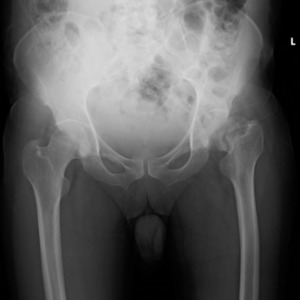
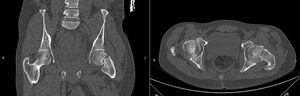
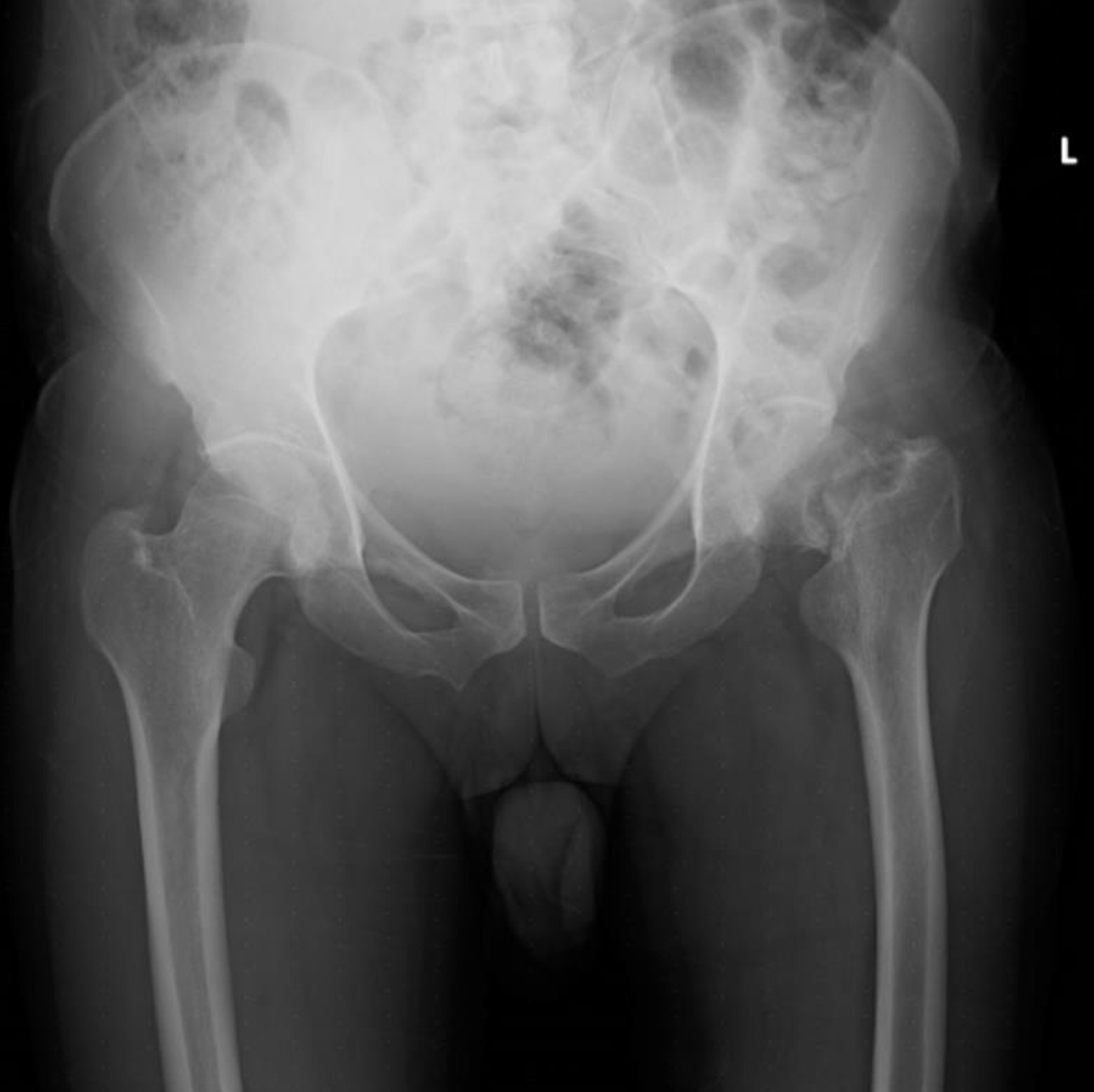
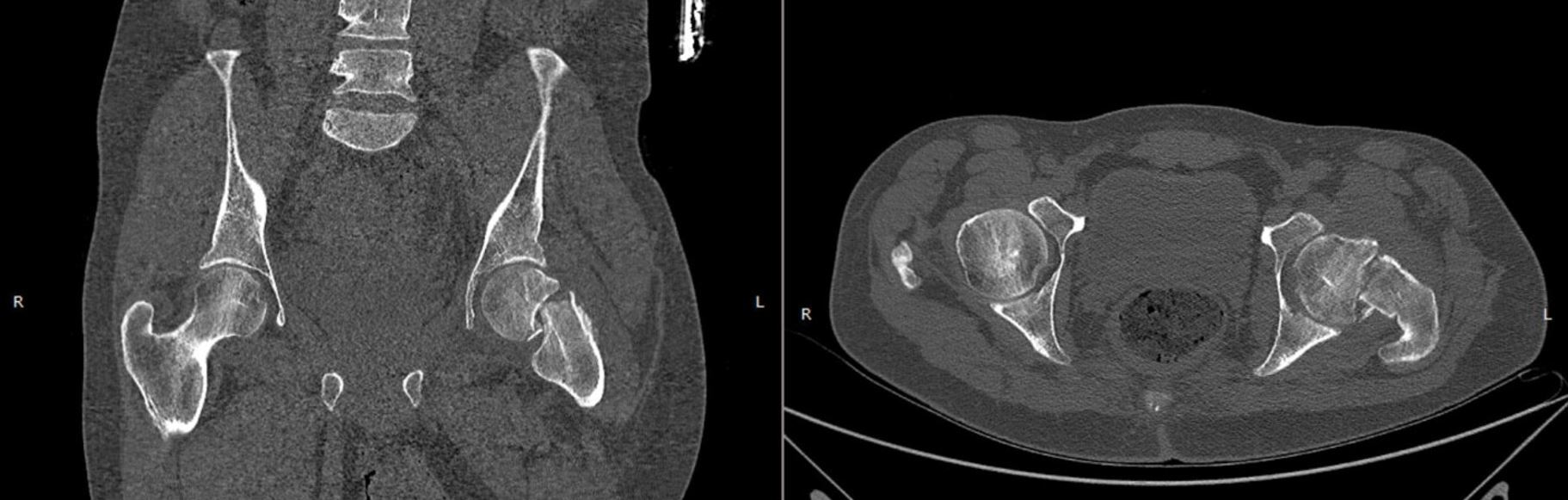
Examination of the patient revealed a shortened and externally rotated left lower extremity and a positive log roll test, characterized by pain elicited with passive internal and external rotation of the hip. Pelvic radiographs showed a displaced left intracapsular femoral neck fracture with a Pauwels angle of approximately 50°, representing a high-shear fracture pattern at the upper limit of Pauwels type II, bordering type III, with no radiological evidence of progression toward union (Figure 1). A computed tomography scan of the pelvis confirmed a fracture gap of 6 mm with a Singh index of 3 or 4 (Figure 2). His previous thoracolumbar magnetic resonance image (MRI) showed no neural axis abnormality, thereby excluding structural and compressive causes of myelopathy. Laboratory evaluation showed normal cell counts (hemoglobin 15.3 g/dL, white blood cell count 5.12×10⁹/L, platelet count 363×10⁹/L), serum calcium of 9 mg/dL, creatinine 0.6 mg/dL, and serum urea nitrogen of 16 mg/dL, with a mildly elevated erythrocyte sedimentation rate of 41 mm/hr. Vitamin D assessment was not performed due to financial constraints.
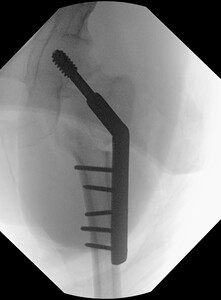
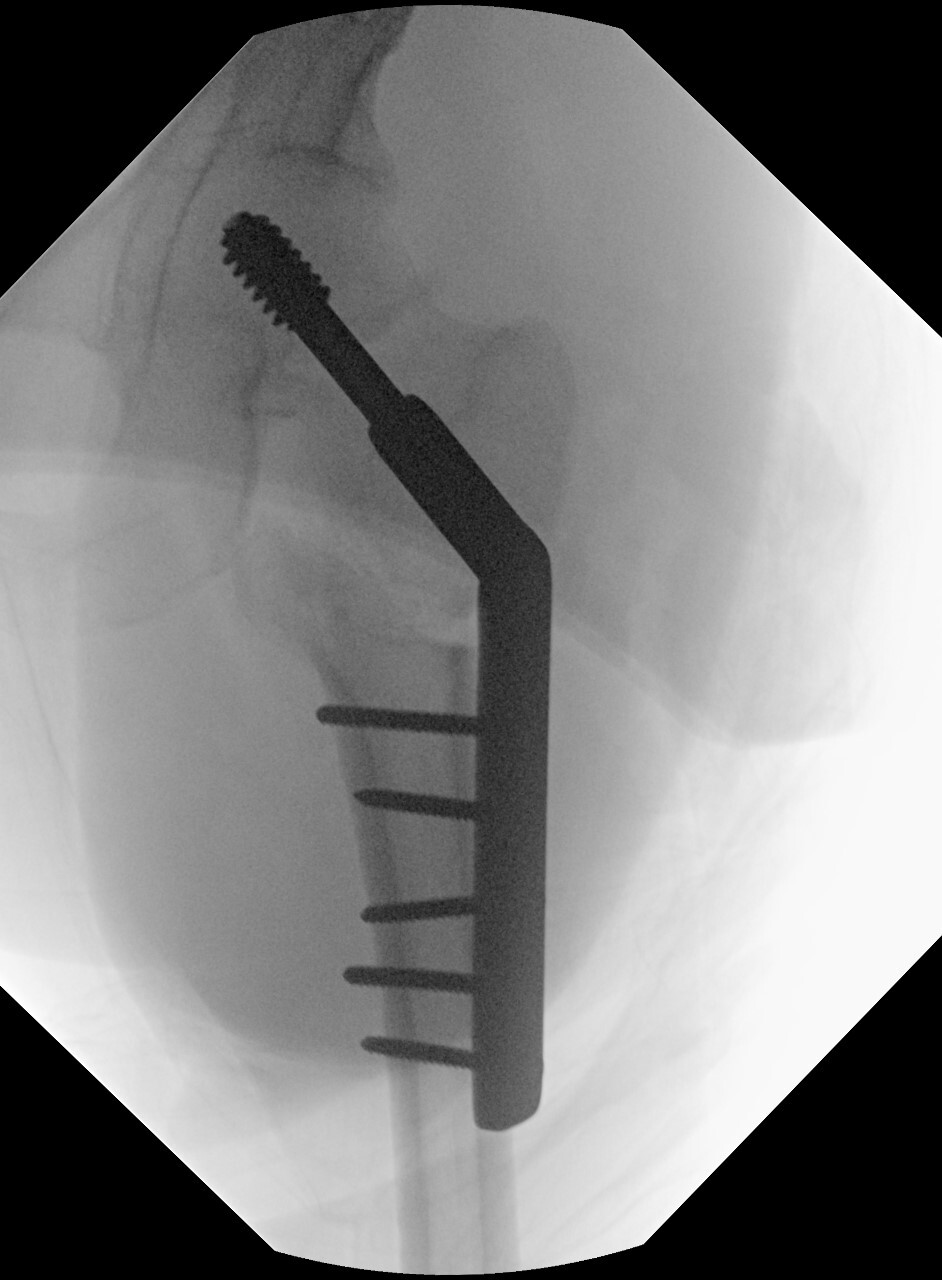
A valgus intertrochanteric osteotomy was planned to promote fracture union by reducing the Pauwels angle and converting shear forces at the fracture site into compressive forces. Intraoperatively, the fracture was restored to length using the Whitman technique and provisionally stabilized with Kirschner wires. A dynamic hip screw was inserted at a pre-calculated angle, and a 20-degree valgus intertrochanteric osteotomy was performed at the level of the lesser trochanter. Valgus correction was confirmed fluoroscopically, and bicortical screws were placed in compression mode to secure the plate (Figure 3).
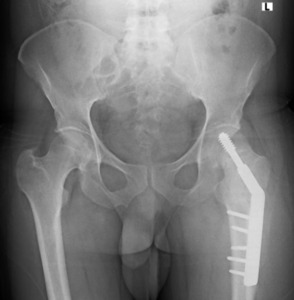
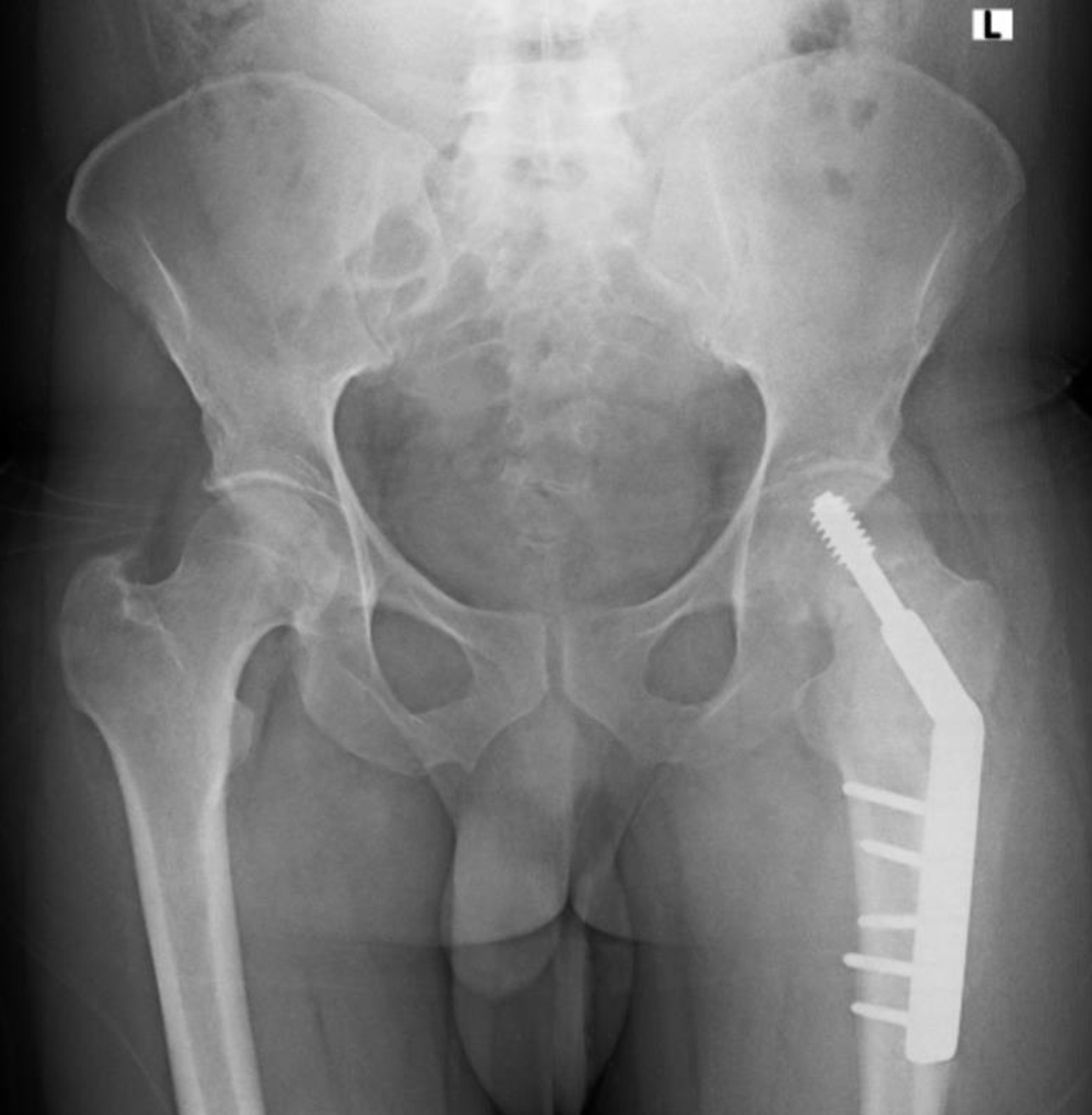
Postoperatively, the patient was kept on toe-touch weight bearing with bilateral crutch support for 6 weeks, then progressed to partial weight bearing until radiographic union was confirmed at 3 months (Figure 4). At the 1-year follow-up, he had returned to his pre-injury functional level (modified Harris hip score of 78), although a mild limp persisted, which required him to use a walking aid for outdoor ambulation. At the 18-month follow-up, he developed a surgical-site infection. It was treated with debridement and plate removal, with subsequent resolution. The patient is satisfied with his current functional status, despite a mild limp.
Discussion
In 1873, Arnaldo Canatani coined the term “lathyrismus,” which Hans Selye later classified into 2 distinct clinical forms.1 The more common form, neurolathyrism, leads to spastic paraparesis and typically presents after 1–6 months of prolonged consumption of grass peas. The rarer form, osteolathyrism, is primarily characterized by absent ossification in late-closing apophyses. In 1997, Haque et al demonstrated that some individuals may exhibit a combination of features from both forms. This is thought to occur due to the presence of 2-cyanoethyl-isoxazolin-5-one in L. sativus seedlings, which can be metabolically converted to BAPN.8 While features of osteolathyrism have been observed in individuals with neurolathyrism, our literature review found no published reports of low-energy femoral neck fractures in this population.
In this case report, we describe a young adult with neurolathyrism who sustained a low-energy femoral neck fracture. The diagnosis of neurolathyrism had been established clinically in childhood on the basis of long-standing, nonprogressive spastic paraparesis in a patient from an endemic area, a history of prolonged grass pea consumption, preserved sensory function, and unremarkable spinal imaging. The injury followed a low-energy mechanism, which is atypical for a displaced femoral neck fracture in this age group. In the absence of radiological features suggestive of other pathological causes, such as primary or secondary bone tumors, the fracture was interpreted as a probable fragility fracture occurring on a background of this pre-existing neurological disorder. This represents a potentially novel clinical observation, the underlying mechanism of which remains unclear. Fracture susceptibility in such patients is likely multifactorial and incompletely understood.
From a mechanical perspective, the spastic paraparesis characteristic of neurolathyrism may predispose patients to falls and abnormal loading patterns due to impaired coordination and altered gait mechanics. In addition to these mechanical factors, experimental evidence supports a role for toxin-mediated impairment of bone quality. BAPN has been shown to increase homocysteine levels, leading to irreversible inhibition of lysyl oxidase (LOX) and disruption of collagen cross-linking.10 This adversely affects key mechanical properties of bone, including tensile strength, toughness, and stiffness, thereby promoting microdamage accumulation and lowering the threshold for fracture. Lees et al (1994) demonstrated dose-dependent deterioration in bone density, sonic velocity, and elasticity following BAPN exposure.11
At the cellular level, BAPN also alters osteoblast function. Turecek et al (2008) reported reduced expression of LOX and osteocalcin following BAPN exposure, while Shen et al (2018) demonstrated decreased bone volume, trabecular thinning and downregulation of genes essential for osteogenesis.12,13 Similarly, Pischon et al (2009) showed that although overall skeletal morphology may appear normal, LOX deficiency results in impaired osteoblast proliferation, reduced differentiation, and defective mineralization, ultimately leading to a structurally weaker bone matrix.14
Taken together, these findings suggest that fracture susceptibility in neurolathyrism may result from the combined effects of abnormal mechanical loading and compromised bone material properties. In our patient, 3 contributing factors are plausible: (1) altered biomechanics and fall risk due to spastic paraparesis, (2) impaired collagen cross-linking reducing bone toughness, and (3) toxin-mediated suppression of osteoblastic activity affecting bone microarchitecture. However, in the absence of bone histology, biochemical markers, and densitometric assessment, the relative contribution of each mechanism cannot be determined. Furthermore, the absence of classic radiographic features of osteolathyrism further tempers any direct causal inference.
Nevertheless, this case highlights an important gap in the literature and underscores the need for further research into the biological and biomechanical factors contributing to fracture risk in lathyrism. A better understanding of these mechanisms may facilitate earlier recognition and inform strategies to optimize skeletal health in affected individuals.
Conclusion
Fragility fractures of the femoral neck in lathyrism are poorly characterized; in our case a valgus intertrochanteric osteotomy produced a good functional outcome. Further research is required to clarify the neurological and toxin-related pathways that may increase fracture susceptibility in this population.
Ethical Approval
No approval was required beyond written informed consent per our institution’s policy for case reports.
Informed Consent
Written, informed consent for submission and publication of this case report, including imaging and associated text, has been obtained from the patients.
Data Availability
All data supporting the findings of this study are included within the article.
Conflict of Interest
The authors declare that they have no conflict of interest.
Funding
None.